More than 11,400 hospitals and surgical centers use regulated, commercially reprocessed “single-use” devices (SUDs), allowing them to save at least 30 to 50% on costs compared to new devices. When health systems use reprocessed SUDs, they also reduce waste, cut greenhouse gas emissions, and enhance supply chain resilience by keeping devices domestically sourced and using them more than once. Note: in the EU, SUD reprocessing is referred to as remanufacturing.
But many health systems are not fully capitalizing on the cost-saving and environmental benefits possible through reprocessing. AMDR has identified the most important steps you can take to create or expand a reprocessing program. Whether you are looking to start a new reprocessing program or optimize your existing one, below are some recommended steps to help you make the most of it.
- Measure the Benefits of Your Reprocessing Program
 Quantifying the savings and environmental benefits of your reprocessing program is crucial. Cost savings can be quickly estimated by multiplying the number of reprocessed devices by at least 30 to 50% of their original cost, depending on negotiated prices. Larger devices, like transfer mats offer even waste reduction, which translates to additional savings. Use tools like our free CO2 emissions reduction calculator – as well as a large, growing body of research and advanced life cycle assessments – to ensure your program is supported by clear data and measurable benefits.
Quantifying the savings and environmental benefits of your reprocessing program is crucial. Cost savings can be quickly estimated by multiplying the number of reprocessed devices by at least 30 to 50% of their original cost, depending on negotiated prices. Larger devices, like transfer mats offer even waste reduction, which translates to additional savings. Use tools like our free CO2 emissions reduction calculator – as well as a large, growing body of research and advanced life cycle assessments – to ensure your program is supported by clear data and measurable benefits.
- Understand Your Reprocessing Partners
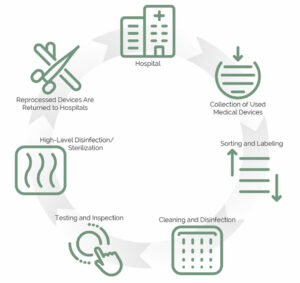
Engage with reprocessors who align with your hospital’s systems and schedules. A good reprocessor will seamlessly integrate with your existing inventory management, purchasing, and collection processes, ensuring that your program runs smoothly. Transparency in how devices are collected, cleaned, and tested is critical to maintaining high standards and trust in reprocessed devices. Consider arranging a tour of a reprocessing facility to see these practices firsthand. Show them your Reprocessing Policy (see box above) and make sure they agree to it.
- Commit to Sustainable Procurement

Sustainable procurement is key to reducing greenhouse gas emissions and waste. Use resources like Practice Greenhealth’s Sustainable Procurement Guide to assess the environmental impact of your supply chain and other Scope 3 emissions. Set ambitious collection goals: even if 100% collection is not feasible, research has shown that high rates of collection significantly increase the effectiveness of a reprocessing program. Continuous improvement and regular auditing of your program will help maintain its effectiveness and alignment with your sustainability goals.
- Simplify Collection Processes
Simplify the collection of used SUDs by having your reprocessing partner train staff on proper procedures and ensuring that collection containers are easily accessible. Collaborate with your reprocessing partner to identify the most effective collection strategies. Small changes, like reducing the size of red sharps bins in operating rooms, can significantly improve collection compliance and the overall efficiency of your program.
- Engage With Staff
 Secure buy-in from leadership and clinicians by emphasizing the financial and environmental benefits of reprocessing. Clear communication about the importance of reprocessing in reducing costs and emissions can drive compliance. Remind staff that exhaustive evidence shows no increased risks from reprocessed devices, and that in fact, reprocessed devices appear to fail less often than original ones. Regularly update staff on program successes and incorporate feedback to maintain engagement. As most healthcare workers want their workplaces to do something about climate change, underscore that through reprocessing, they are personally contributing to the solution.
Secure buy-in from leadership and clinicians by emphasizing the financial and environmental benefits of reprocessing. Clear communication about the importance of reprocessing in reducing costs and emissions can drive compliance. Remind staff that exhaustive evidence shows no increased risks from reprocessed devices, and that in fact, reprocessed devices appear to fail less often than original ones. Regularly update staff on program successes and incorporate feedback to maintain engagement. As most healthcare workers want their workplaces to do something about climate change, underscore that through reprocessing, they are personally contributing to the solution.
- Advocate for Continuous Improvement
Regularly audit your sustainability program and advocate for new science and technology that can further reduce emissions and costs. If your organization lacks a sustainability officer, consider creating this role to drive green decision-making. Use visual data to communicate the impact of your reprocessing program, both internally and externally, to build broader support.
Additional External Resources
- Maximize your reprocessing program
- Transparency in reprocessing: A healthcare provider’s checklist
- Profitable, sustainable reprocessing programs: A healthcare provider’s checklist
- Patient safety in reprocessing: A healthcare provider’s checklist
